 For example: while the electron notation of the element carbon is 1s☢s² 2p², the Noble Gas notation would be 2s☢p². The noble gases can be used in conjunction with the electron configuration notation to make what is called the Noble Gas Notation. Xenon is used in xenon arc lamps, and it has anaesthetic properties. 
Krypton is also used in lasers, which are used by doctors for eye surgery. Helium and argon are commonly used to shield a welding arc, and the surrounding base metal from the atmosphere during welding. Helium, due to its nonreactivity (compared with flammable hydrogen) and lightness, is often used in blimps and balloons. Some of the noble gases glow distinctive colors when used inside lighting tubes ( neon lights). Argon is often used as a suitable safe and inert atmosphere for the inside of filament light bulbs, and is also used as an inert atmosphere in the synthesis of air and moisture sensitive compounds (as an alternative for nitrogen). One of the most commonly encountered uses of the noble gases in everyday life is in lighting. This is why they are all monatomic gases under normal conditions, even those with larger atomic masses than many normally solid elements. The noble gases have very weak inter-atomic forces of attraction, and consequently very low melting points and boiling points. 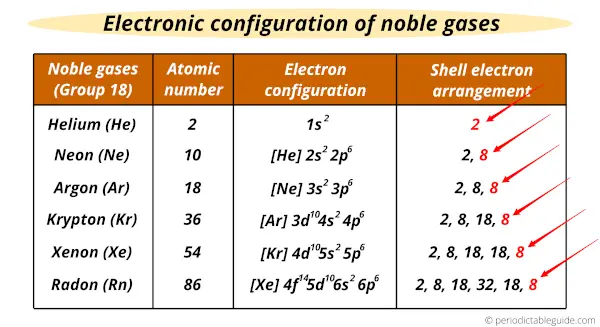
The noble gases have high ionization energies and negligible electronegativities. They have little tendency to gain or lose electrons. The noble gases' lack of reactivity can be explained in terms of them having a "complete valence shell". Recently argon compounds have also been successfully characterised. No conventional compounds of helium or neon have yet been prepared, while xenon and krypton are known to show some reactivity in the laboratory. Īll of them exhibit an extremely low chemical reactivity and very few noble gas compounds have been prepared. According to atomic theory derived from quantum mechanics and experimental trends, atoms with full valence electron shells are extraordinarily stable and therefore do not form chemical bonds. Valence electrons are the outermost electrons of an atom and are normally the only electrons which can participate in chemical bonding. Noble gases have full valence electron shells. Under standard conditions, the noble gases all occur as monatomic gases. The isolation of helium on Earth had to wait until 1895. Helium was detected spectrographically in the Sun in 1868. Eventually, all the known noble gases except for helium were discovered in the air, with argon being much more common than the others, and the table was completed. With this discovery, they realized that a whole class of gases was missing from the periodic table. Along with scientist William Ramsay, Lord Rayleigh theorized that the nitrogen extracted from air was associated with another gas, argon. In the late nineteenth century, Lord Rayleigh discovered that some samples of nitrogen from the air were of a different density than nitrogen resulting from chemical reactions. The existence of noble gases was not known until after the advent of the periodic table. 
Another old term is rare gases, although argon forms a fairly considerable part (0.93% by volume, 1.29% by mass) of the Earth's atmosphere. The noble gases have also been referred to as inert gases, but these terms are not strictly accurate because several of them do take part in chemical reactions. This refers to the extremely low level of reactivity under normal conditions. "Noble gas" is the translation of the German Edelgas, which was in use as early as 1898. The seven noble gases are: helium, neon, argon, krypton, xenon, radon, and ununoctium. Noble gases have numerous important applications in lighting, welding and space technology. 
Each of them has its melting and boiling point close together, so that only a small temperature range exists for each noble gas in which it is a liquid. Under normal conditions, they occur as odorless, colorless, monatomic gases. Noble gases rarely react with other elements since they are already stable. A thorough explanation requires an understanding of electronic configuration, with references to quantum mechanics. Chemically, the noble gases are very stable due to having the maximum number of valence electrons their outer shell can hold. The group is also called the helium family or neon family. The noble gases are the elements in group 18 (also sometimes Group 0 IUPAC Style, or Group 8) of the periodic table. File:Electron shell 010 Neon.svg Neon, like all noble gases except helium, has a "full" (eight electron) valence (outermost) electron shell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
